Carbon Sp3 Electric Configuration Diagram 62.chemical Bondin
Em química orgânica, não conseguir entender o que faz Solved how many sp3 hybridized carbon atoms are in this Sp2 hybridization shape
Hibridación: Estructura de la Química Orgánica Acetileno | MCC | Radio
Sp3 hybridization orbitals sp2 2p energies stability increasing Ch 2: sp3 hybridisation Homework smelly science do
How does carbon hybridize its s and p orbitals and what effect does
What does sp2 and sp3 hybridization mean? + exampleWhat is an sp3 hybridized carbon atom Carbon electric configuration diagramHybridization of atomic orbitals.
What is hybridization?- sp3, sp2, examples and formulaSp3 hybridization part 02 Hybridization sp figure sp3 orbitals hybrid diamond structure tetrahedral process geometry cubic globalsino emSp hybridization carbon.

Solved 7. for the structure shown, label each carbon as sp3,
Hybrid orbitals1: schematic representation of hybridised carbon in sp 3 -, sp 2 -and Sp3, sp2 and sp hybridization, geometry and bond anglesSolved 10. how many sp3 carbons are in the structure given.
Hybridisation sp3 ch2 electron 2p 2s promoteSp3d orbitals Sp3 hybridizationStructures of diamond comprised of sp3 carbon atoms and of.

Carbon sp3 electric configuration diagram
62.chemical bonding (9)- covalent bonding(8)- sp3 hybridizationSp3 hybridization electron geometry diagram Hibridación: estructura de la química orgánica acetilenoHybridization sp3 bonding methane chemical covalent electron orbital hydrogen atoms thus overlap.
Solved how many sp3 hybridized carbons does this structureHybridization of co2 Sigma bonds come in six varieties: pi bonds come in one – masterCarbon hybridization orbital diagram.

Sp3 hybridization
Bonds sigma pi sp2 sp3 sp hybridization count varieties chemistry orbitals thereforeSolved the structure shown has sp3 carbons, sp2 carbons and What are sp sp2 sp3 orbitals exampleCarbon hybridization orbital diagram.
Hybridization: sp, sp2, sp3 & sp3d atomic orbitals, properties & examplesThe smelly science homework blog: homework due 10.8.14 What is the geometry of: (a) an sp3 hybridized carbon atom? (b) an sp2.


Hibridación: Estructura de la Química Orgánica Acetileno | MCC | Radio

What is Hybridization?- sp3, sp2, Examples and Formula

Ch 2: sp3 hybridisation

Sp Hybridization Carbon
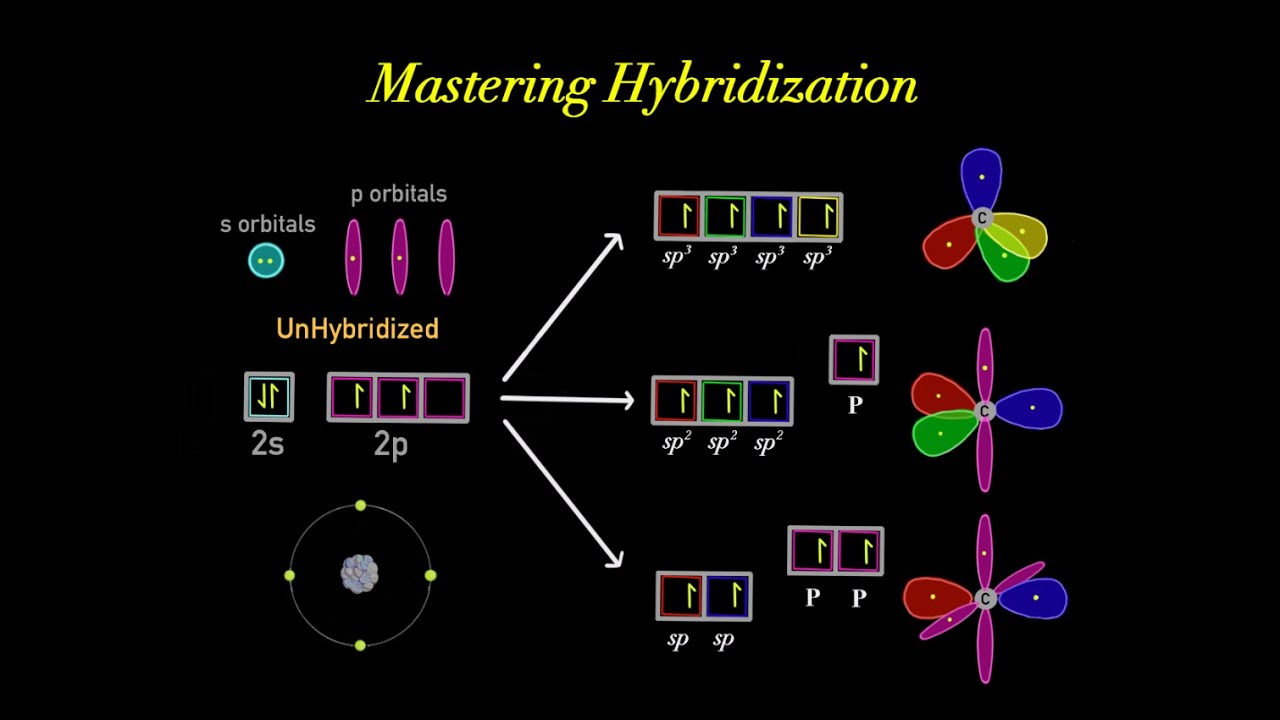
Carbon Hybridization Orbital Diagram

Hybrid Orbitals

Structures of diamond comprised of sp3 carbon atoms and of

What is the geometry of: (a) an sp3 hybridized carbon atom? (b) an sp2